As 2020 came to an end, the world was overjoyed that the year was over. Unfortunately, many of the problems that arose during 2020 didn’t end with the year. This includes the COVID-19 pandemic that is raging through many countries with little hope that it will be ending anytime soon. The majority of the world masked up, washed their hands, kept their distance, and isolated themselves after potentially risky encounters. While it is proving to be very controversial, a few different pharmaceutical companies have developed a vaccine in order to combat infection of the virus in hopes to eventually create a general immunity to the virus. Let’s talk about what the vaccine is, how it works, and the risks. I have interviewed a few people who have already received the vaccine in order to educate others about what the experience was like and how they felt afterward.

The Four W’s of the Vaccine
Who produced the COVID-19 vaccine? Two different pharmaceutical companies have produced an FDA approved vaccine to fight the virus. The Pfizer-BioNTech created the first vaccine. On the CDC’s page surrounding the Pfizer-BioNTech vaccine, it has a list of common allergens that are NOT included in the vaccine. It is recommended to people about the age of sixteen years old. It also includes information on the shot and how it is administered. This vaccine consists of two doses that are twenty-one days apart. The other company that has developed a vaccine is Moderna. This page also includes a list of common allergens that the vaccine does not include. One major difference is the age group the vaccine is recommended for. Instead of being recommended for anyone sixteen or up, the Moderna vaccine is recommended for people over eighteen. Another difference is in the days in between shots. The Moderna vaccine’s second dose is administered “one month (28 days) apart.” (CDC).
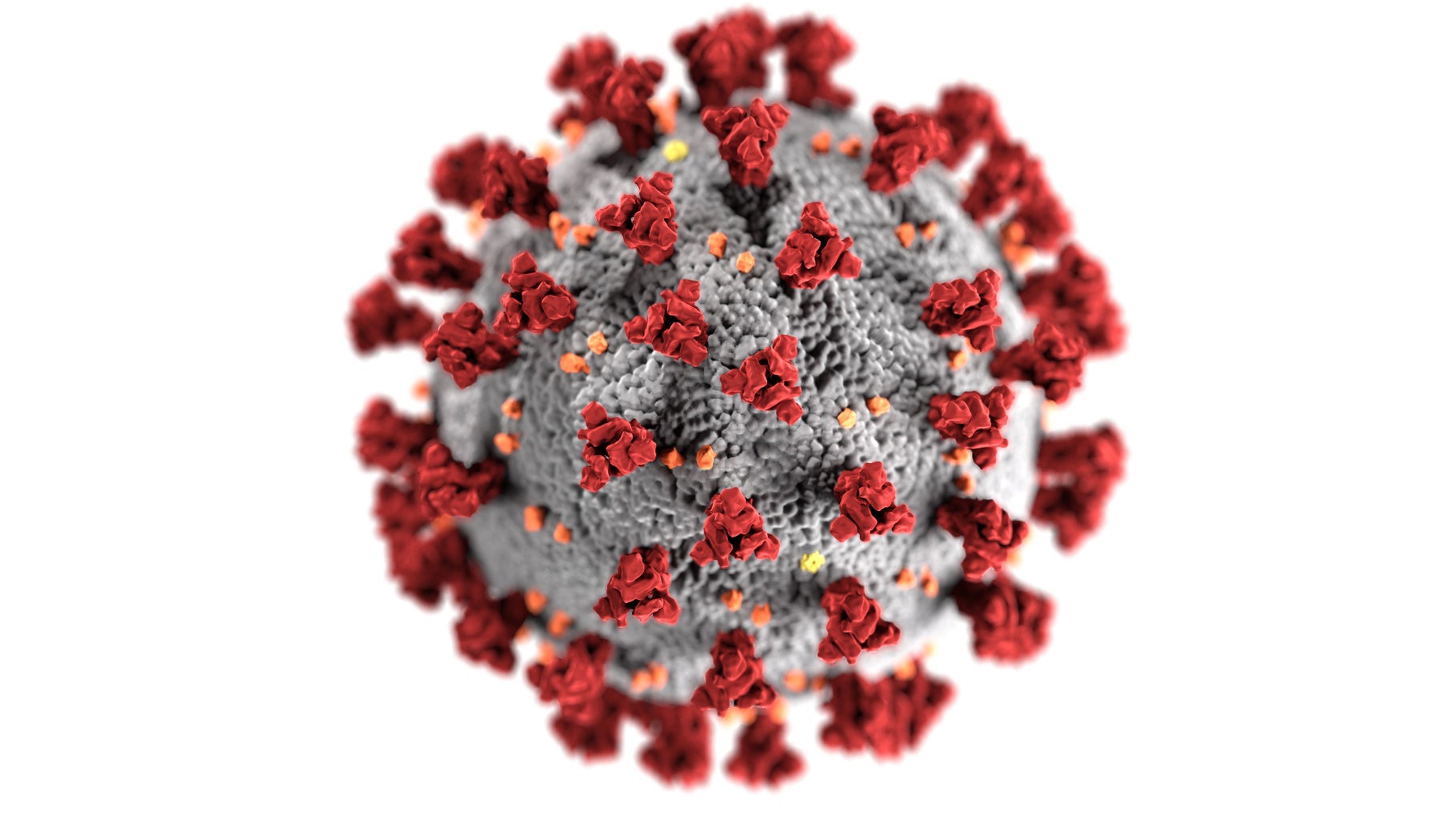
What is the COVID vaccine? The CDC describes the COVID-19 vaccine as a “messenger RNA or mRNA” (CDC), which means that the vaccine uses mRNA to create a protein that your immune systems then respond to. When the immune system recognizes this protein, it produces antibodies that are able to protect us from being infected by the virus in the future. Specifically, with the COVID-19 vaccine, that protein is known as the “spike protein… [which] is found on the surface of the virus that causes COVID-19” (CDC). The red parts of the picture below are the spike proteins.
What is the success rate? Both vaccines have an “efficacy rate of approximately 95%,” (Biospace). The Pfizer-BioNTech trials revealed that out of everyone included in their clinical trial, less than 200 people total contacted COVID-19 after receiving the vaccine, “162 of them occurring in the placebo,” (Biospace) patients. Similarly, to the Moderna vaccine, after receiving the vaccine, “only eleven [cases] compared to the 185 cases in the placebo,” (Biospace) patients. On top of that, less than two percent of patients have had harmful negative reactions.
Real-Life Application of the Vaccine
The COVID-19 pandemic has taken the world by storm. Since the vaccine’s approvals in the US and in many other countries, there have been “more than 32.4 million doses,” (Bloomberg) administered. Roughly 10 million have been given in the US alone. The distribution of the vaccine in the US is based on the risk of an individual. It started with healthcare personnel and people in long term care facilities. These people are deemed more at risk due to their exposure levels and their ability to fight off the virus. The second wave will administer vaccines to people who are frontline and/or essential workers and people who are over seventy-five years old. As more vaccines are created more and more people will have access to the vaccine. By getting the vaccine, people are creating a greater chance for herd immunity. “Herd immunity is a term used to describe when enough people have protection—either from previous infection or vaccination—that it is unlikely a virus or bacteria can spread and cause disease,” (CDC) the number of people that will need to receive the vaccine to create this immunity is unknown. But by more people getting it, we get closer to that point.

Heidi is a Mental Healthcare Worker. “I chose to get be vaccinated because I have not seen my elderly parents for the last ten months due to the social distance protocols.” She has received both doses of the vaccine and told me about both experiences. She walked into the clinic, checked in, received a symptom check, and then was called back for the injection. She also had to wait for fifteen minutes after injection to make sure that there were no adverse effects. “The first shot was not painful at all and my arm wasn’t affected. The second was moderately painful and I did have a great deal of muscle pain in my arm for about an hour,” she continued with “on a scale of one to ten, I would say the first one was a one and the second one was a five.” With the first injection, she felt zero side effects. She felt healthy and normal. After receiving the second one, she said that she had muscle pain in her arm and chest for a few days with a headache on top, but eventually they subsided, “by the third day after the second injection I was back to work and fine.” After a few days of pain, she is not only protected from contracting COVID-19. She is now able to help her clients in a much safer environment.

Sources:
https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/mRNA.html
https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/Pfizer-BioNTech.html
https://www.biospace.com/article/a-timeline-of-covid-19-vaccine-development/
https://www.bloomberg.com/graphics/covid-vaccine-tracker-global-distribution/