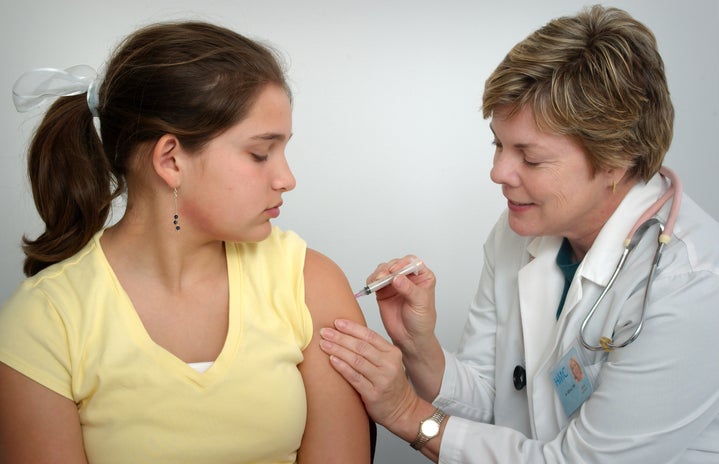
On Monday, Nov. 9, Pfizer, a major American pharmaceutical company, and their German partner company BioNTech announced the first results of their attempts to create an effective vaccine. This comes on the heels of national statistics reporting new spikes in coronavirus case numbers. Initial results indicate that the vaccine would be 90 percent effective. Despite the good news, a vaccine will not be ready in time to save 2020. In fact, it may not be available to the general public until the middle of next year. This announcement is just the beginning.
Trials for this potential vaccine began in limited trial runs in May to test for safety concerns and effectiveness. July is when the true large-scale vaccine trials began. The clinical study was expansive, with over 44,000 participants. Half were given a placebo, while the other half were given the vaccine. Only the independent board of experts knew the ratios of vaccine dispersion among the participants—this is due to the results being “blinded.” People in the study will be monitored for the next two years. Currently, both companies are reporting no serious health or safety risks associated with the new vaccine. If it gets emergency approval from the FDA, Pfizer and BioNTech could produce 30 to 40 million doses. Once approved and on the market, the first ones to receive the vaccine would be groups at high risk or health care workers.
Furthermore, even with a 90 percent effectiveness rate, that still leaves one in 10 people vulnerable to the virus. The announcement for this potential vaccine does not guarantee the end of social distance protocols or mask-wearing due to the vulnerable population. It may take a while to become widely available. Half of Americans say they would take the vaccine. On the Federal Drug Administration’s part, transparent communication to the public about the vaccine and its pros and cons is key to being able to successfully distribute the doses effectively. The more Americans that are willing to take the vaccine, the less likely it is to see case numbers rise.
Overall, these results from Pfizer and BioNTech seem to be very promising. This is the first time a pharmaceutical company has gotten this far with this much success for a COVID-19 vaccine. Although it will take time and patience to implement not only to the American public but the world, there is hope on the horizon for getting coronavirus case numbers down in the future. This is major progress in the ongoing fight against this disease.
Want to see more HCFSU? Be sure to like us on Facebook and follow us on Instagram, Twitter, TikTok, Youtube and Pinterest!